 |
 |
- Search
| Neurointervention > Volume 17(2); 2022 > Article |
|
Abstract
Purpose
Materials and Methods
Results
Notes
Fund
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIT) (No. 2018R1A2B6003143). The authors have no relevant financial or non-financial interests to disclose.
Ethics Statement
Study permissions from the Institutional Review Board (IRB no. 2021-0891) and informed consent from each patient for the taurine challenge test and publication were obtained.
Conflicts of Interest
DCS has been the Editor-in-Chief of the Neurointervention since 2018. No potential conflict of interest relevant to this article was reported. YS has been the Assistant Editor of the Neurointervention since 2019. No potential conflict of interest relevant to this article was reported. No other authors have any conflict of interest to disclose.
Fig. 1.

Fig. 2.
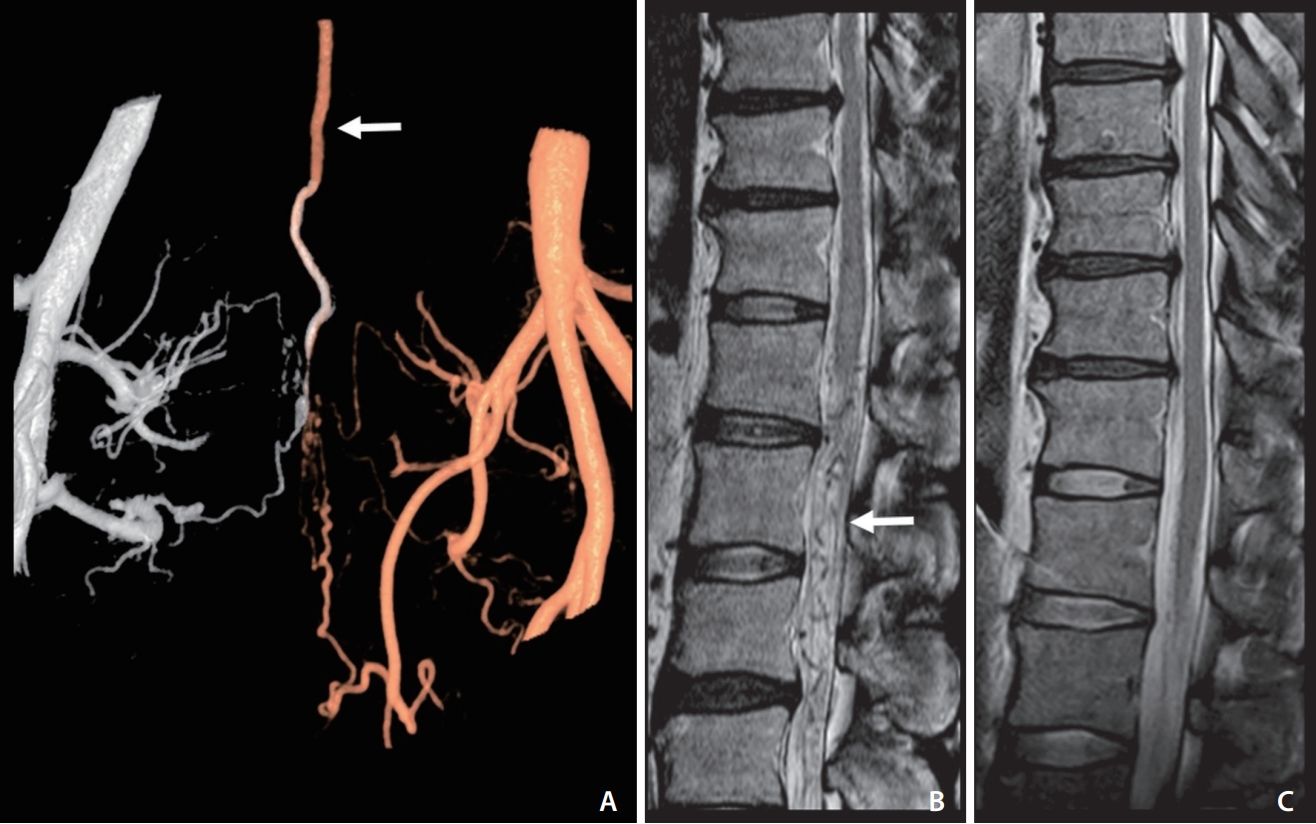
Fig. 3.
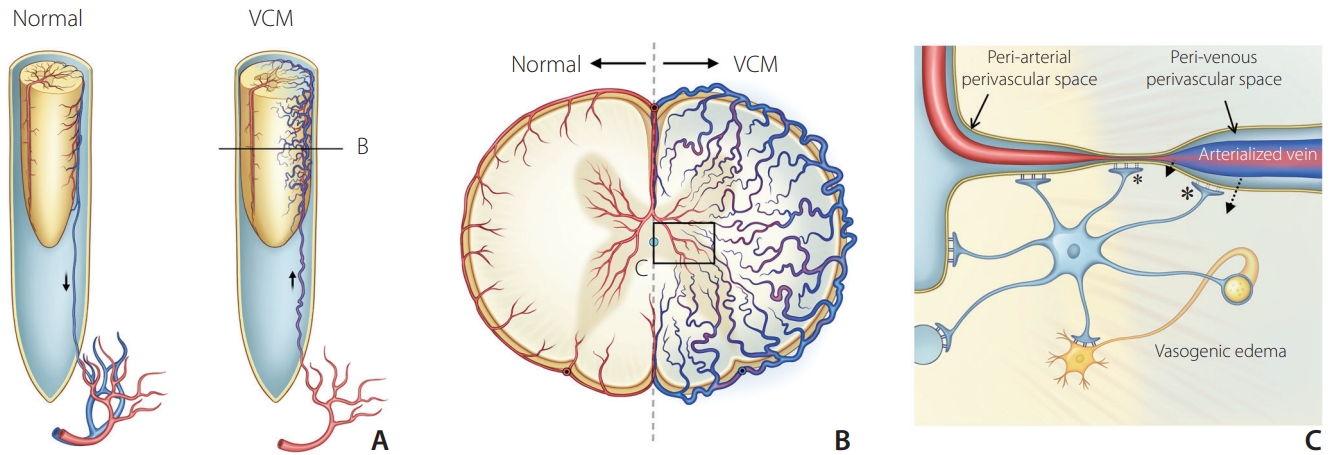
Table 1.
| Case | Age/Sex | Dx. | Lesion level | Steroid* | Taurine-rich foods | Pre-PSMS | Post-PSMS | Pre–Post PSMS† | Taurine challenge after Tx. | Free taurinerich food intake | F/U (mo) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 42/M | SDAVF | S2 | Yes | Webfoot octopus, squid, small octopus, Bacchus® 2,000 mg | 3 | 1 | 2 | Done at 24 months | Yes | 24 |
| 2 | 71/M | SDAVF | T10 | NA | Webfoot octopus, squid, small octopus, crab, scallops | 4 | 2 | 2 | Done at 26 months | Yes | 26 |
| 3 | 63/F | SEDAVF | L2 | NA | Bacchus® 1,000 mg | 4 | 1 | 3 | Done at 48 months | Yes | 48 |
| 4 | 45/M | SDAVF | L1 | No | Bacchus® 2,000 mg | 5 | 0 | 5 | Done at 76 months | Yes | 76 |
Bacchus® (Dong-A Pharmaceutical, Seoul, Korea).
VCM, venous congestive myelopathy; Dx., diagnosis; PSMS, pain-sensory-motor-sphincter score; Tx., treatment; F/U, follow-up; M, male; F, female; SDAVF, spinal dural arteriovenous fistula; SEDAVF, spinal epidural arteriovenous fistula; NA, not available.







